In this pictorial essay,
we will illustrate the computed tomographic appearances of extranodal lymphoma in the abdomen including AIDS-related lymphomas and highlight points to differentiate them from its mimics.
We classify extranodal lymphoma according to the site involved into: (a) solid abdominal organs or (b) hollow organs of the gastrointestinal tract(GIT).
Solid organ involvement:
Spleen and liver are the commonly affected organs followed by kidney,
pancreas and adrenal glands.[1,2,3] Lymphomatous involvement of the solid organs may occur as a focal,
multifocal,
or diffuse disease process.
In focal and multifocal disease,
discrete solid nodules are present while in diffuse disease,
there is uniform infiltration of the involved organ.[4]
Spleen:
Spleen is considered to be a nodal organ in HD and an extranodal organ in NHL.[1,2,4] Primary involvement of spleen is rare while it is secondarily involved in 20–40% of the patients.[4]
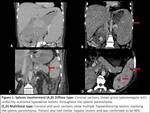
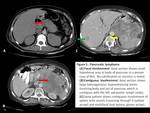
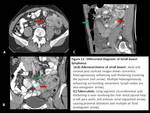
Diffuse infiltration is the most common form of splenic lymphoma.[1,2,4] On CT,
spleen may be normal or enlarged in size while few cases may show ill defined hypoattenuating nodules measuring > 1cm and scattered throughout the splenic parenchyma after intravenous contrast administration.[2,4] Marked splenomegaly almost always indicates infiltration though mild to moderate splenomegaly can be due to reactive process in other cases of lymphoma.[2,3,4] FDG/PET CT is more specific for detection of diffuse infiltration in absence of above findings.[1]
Focal or multifocal involvement of spleen is rare and shows small homogeneous hypoattenuating nodules of varying sizes scattered in the splenic parenchyma with associated bulky coalescent hilar lymph nodes.[1,3,4]
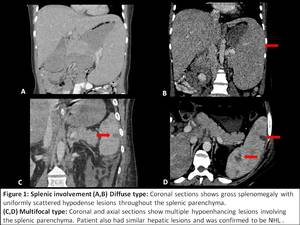
Fig. 1: Splenic lymphoma
Differential diagnosis includes:
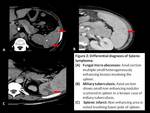
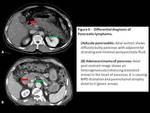
Fungal micro-abscesses: Nodules are smaller sized and show heterogeneous post contrast enhancement.
[2,3,4]
Miliary tuberculosis: Smaller multiple hypodense nodules showing no or minimal post contrast enhancement.
Splenic infarction: Involved area is non enhancing as compared to hypoenhancement of lymphoma.[2]
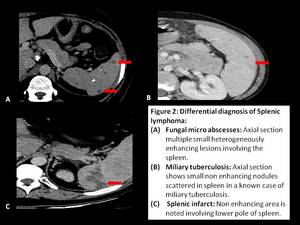
Fig. 2: Differential diagnosis of Splenic lymphoma
Liver:
Primary hepatic involvement is rare and is seen in immunocompromised patients.[1,4] However,
secondary hepatic involvement is fairly common and is seen in up to 15% of patients.[2,4] Primary and secondary lymphomas of liver are commonly NHL rather than HD.
HD of the liver is almost invariably associated with disease of the spleen while NHL of liver can occur without splenic involvement.[2,3,4]
Pattern of involvement is same as that of spleen: diffuse,
focal or multifocal types,
with diffuse type being the commonest.[1,3,4] Fig. 3
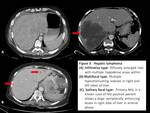
Diffuse pattern may show hepatomegaly,
a nonspecific finding along with small diffusely infiltrated hypoattenuated nodules which can easily be overlooked on cross sectional imaging.[4]
Focal or multifocal nodular type is seen in about 10% of cases.
They appear as small well circumscribed hypo-attenuated lesions which show homogeneous and hypo-enhancement as compared to rest of the liver.[2,3]
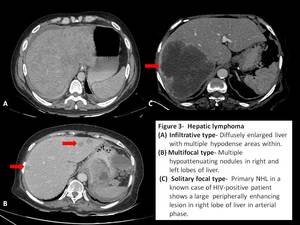
Fig. 3: Hepatic lymphoma
Differential diagnosis includes:[1,2,4]
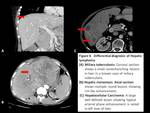
Miliary tuberculosis: Lesions are smaller and show no or minimal post contrast enhancement.
Hepatic metastasis: Variable sized,
multiple heterogeneous lesions with rim like or target type of enhancement,
and a history of known primary lesion.
Hepatocellular carcinoma: They may show typical arterial phase enhancement and rapid washout in the background of cirrhotic liver.
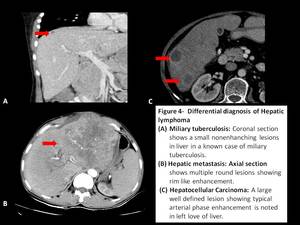
Fig. 4: Differential diagnosis of Hepatic lymphoma
Pancreas:
Pancreatic involvement is seen in about 30% of cases of NHL,
usually from contiguous nodal infiltration.[2,4] Pancreatic HD is extremely rare.
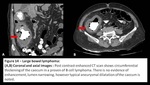
Two patterns of involvement can be seen: Circumscribed focal mass or diffuse glandular enlargement.[1,4]
Circumscribed focal mass can occur in any part of pancreas.
They appear as well defined,
hypodense lesions showing homogeneous enhancement with minimal or no ductal obstruction.
Necrosis and calcification are usually not seen.[3,4]
Diffuse glandular infiltration appears as diffuse enlargement of the gland with infiltration of the peripancreatic fat mimicking acute pancreatitis.[1,3] Low attenuation of the affected gland with reduced but homogeneous post contrast enhancement is the usually presentation.
Encasement of vessels without invasion is another specific feature of lymphoma.
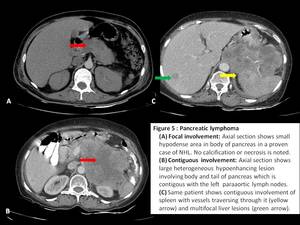
Fig. 5: Pancreatic lymphoma
Differential diagnosis includes:[1,2,4]
Acute pancreatitis: Can be differentiated based on clinical presentation or imaging features of peripancreatic free fluid,
necrosis or presence of complications such as ascites or pleural effusion.
Adenocarcinoma of pancreas: It presents as an ill defined,
heterogeneously enhancing mass lesion with areas of calcification and vascular invasion.
Extensive ductal dilatation and pancreatic atrophy distal to tumor are other important distinguishing features.
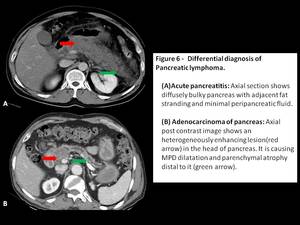
Fig. 6: Differential diagnosis of Pancreatic lymphoma
Kidney:
Kidney is the most commonly involved part of the GU tract.[1] Renal involvement occurs in 3–8% of patients with lymphoma.
Primary involvement is rare and secondary involvement due to hematogenous spread or direct extension of disease from contiguous retroperitoneal lymphoma is observed in late-stages of lymphoma.[2,3]
Patterns of renal involvement include multiple circumscribed masses,
direct infiltration from adjacent nodes,
a solitary mass,
an isolated perinephric mass and diffuse infiltration.[1]
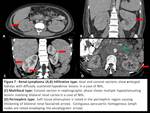
Multiple focal lesions is the most common presentation seen in 60% of the cases.
Findings include small,
homogeneous,
hypodense lesions (1-3cm) which enhance less than the renal parenchyma after intravenous contrast administration.[4]
Diffuse infiltration is seen in 20% of cases and is always bilateral.
Nephromegaly due to proliferation of the renal interstitium with maintained renal contour is the primary finding.[2,4] Multiple poorly enhancing areas in renal parenchyma following intravenous contrast administration is seen in minority of cases.[1,4]
Contiguous renal invasion from retroperitoneal lymph nodes is seen in 25% of cases and typically manifests as a bulky mass which envelopes but does not obstruct the renal vessels.
It may invade the hilum and displaces the affected kidney.
[2,4]
Perirenal involvement presents as a homogeneous soft tissue density lesion encompassing the renal parenchyma,
without causing significant renal invasion.
In milder cases,
there is just thickening of the renal fascia or presence of nodules in the perirenal space.[3,4]
Ureter and urinary bladder are seldom involved primarily with most of the cases due to contiguous spread from adjoining disease process.[2]
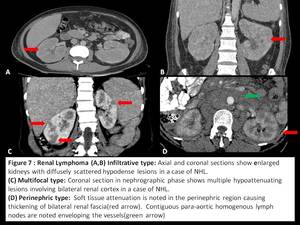
Fig. 7: Renal lymphoma
Differential diagnosis includes:
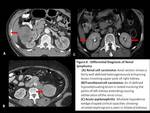
Renal cell carcinoma: Appears as ill defined heterogeneous soft tissue lesion with varying areas of necrosis,
hemorrhage or calcifications and rapid post contrast enhancement,
features which are atypical for lymphoma.[1,2,4]
Transitional cell carcinoma: A soft tissue density mass centered in the renal pelvis and showing mild post-contrast enhancement with obliteration of renal sinus producing “faceless kidney”.[6]
Acute pyelonephritis: Multiple hypodense wedge shaped cortical opacities showing striated nephrogram pattern in excretory phase of CT is diagnostic of pyelonephritis.[7]
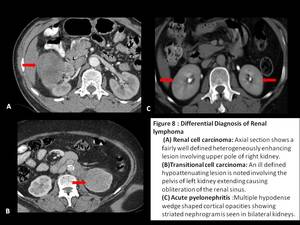
Fig. 8: Differential diagnosis of Renal lymphoma
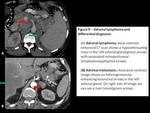
Adrenal gland:
Adrenal lymphoma is rare and occurs in 4% of all cases of NHL with bilateral involvement in 50% of these cases.[1,4] Imaging findings are non-specific and can consist of well-defined homogeneous masses,
small nodules,
or diffuse increase in the size of the glands.[2,3] In addition,
retroperitoneal adenopathy or other extranodal lesions can be identified.[4]
Differential diagnosis includes:
Adrenal adenoma: Found incidentally as small focal lesions with HU <10 in 70% cases( lipid rich adenomas) and 60% absolute and 40% relative washout after 15 minutes post contrast.[8]
Adrenal metastasis: Variable sized heterogeneously enhancing lesions with a history of primary malignancy can help us identify them.[8]
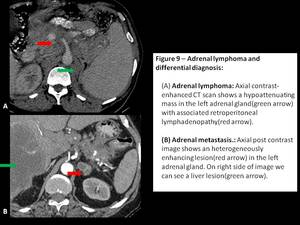
Fig. 9: Adrenal lymphoma and differential diagnosis
Hollow organ involvement:
Extranodal lymphoma in the gastrointestinal tract occurs in 10–30% of all patients with NHL.[1,2] The stomach,
small bowel,
large bowel,
and esophagus can be involved in decreasing order of frequency.[1,2]
It may originate and extend along the submucosa or the deep mucosal layer,
unlike adenocarcinoma which arises from the mucosa.
Because of this pattern of extension,
it can be easily missed on endoscopy.
Hence CT is the primary modality for evaluating and staging of the gastrointestinal lymphoma.[4]
Pattern of involvement include: nodular,
polypoid,
infiltrative,
aneurysmal or cavitary,
ulcerative,
and mixed types.
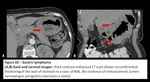
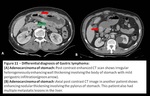
Stomach:
Lymphoma accounts for only 3%–5% of all malignant gastric neoplasms.
NHL is about ten times more common than HD among gastric lymphomas.[4] In addition,
secondary gastric lymphoma is more common than primary lymphoma.
There is no site predilection with all portions of the stomach being equally involved.[4]
Low-grade mucosa-associated lymphoid tissue (MALT) lymphoma is the most common type of primary gastric lymphoma accounting for 50-70% of cases and has strong association with Helicobacter pylori gastritis.[1,4] MALT can be regressed with treatment for H.
Pylori or can progress to high grade B-cell NHL.[4]
On imaging,
there can be extensive/diffuse infiltration,
focal thickening of the gastric wall (>3cm),
circumferential parietal involvement or polypoidal growth.[1,4] It is difficult to distinguish the grades of lymphoma on imaging,
however following features favor its diagnosis:[4]
(a) diffuse,
extensive,
or severe gastric wall thickening;
(b) minimal or mild tumor enhancement;
(c) preservation of the perigastric fat plane;
(d) no gastric lumen narrowing;
(e) bulky and smooth lymphadenopathy,
which can extend below the renal pedicle.
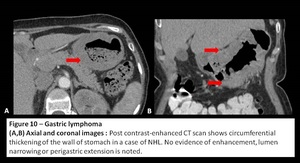
Fig. 10: Gastric Lymphoma
Differential diagnosis includes:
Adenocarcinoma of stomach: It appears as ill defined focal or diffuse heterogeneously enhancing gastric wall thickening which causes mild to severe luminal narrowing and perigastric infiltration.
[4]
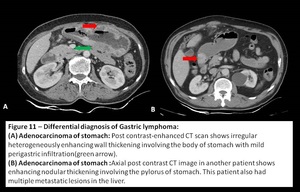
Fig. 11: Differential diagnosis of Gastric lymphoma
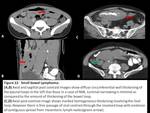
Small bowel:
It is the second most common site of gastrointestinal lymphoma with distal ileum being the most affected segment due to abundant lymphoid tissue.[2,4] It accounts for ~20% of all malignant tumors of the small bowel.[4] In recent years due to B-cell hyperactivation in HIV-positive patients,
its incidence has increased.[2,3] Primary NHL,
Burkitt lymphoma,
MALT-type lymphoma,
and,
rarely,
HD have been found to involve the small intestine.[3]
Pattern of involvement includes circumferential asymmetric wall thickening,
solitary or multiple nodules and contiguous involvement from mesenteric nodes.
Typically one can see aneurysmal dilatation of the affected bowel segment which is absent in adenocarcinoma.[1,2,4]
Bowel perforation is an important but uncommon complication which is seen with T-cell lymphoma,
PTLD,
after chemotherapy or radiation therapy.[1]
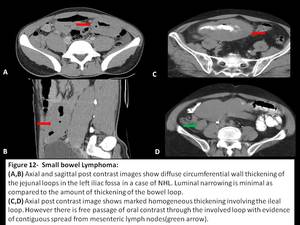
Fig. 12: Small bowel lymphoma
Differential diagnosis includes:
Adenocarcinoma of small bowel: Polypoidal or concentric irregular bowel wall thickening showing heterogenous enhancement.
Lesion affects single bowel segment rather than multiple as in lymphoma and cause significant luminal narrowing and obstruction.
[9]
Tuberculosis: There is circumferential wall thickening of affected segment( commonly IC junction) causing luminal narrowing with dilatation of the proximal bowel segment.
Surrounding necrotic mesenteric lymphadenopathy is also seen.
[10]
Crohn’s disease: Presents as bowel wall thickening (1-2 cm) which is most frequently seen in the terminal ileum with mucosal or mural hyperenhancement and submucosal fat deposition. Presence of skip lesions and discrete ulcers is also typical of Crohn’s disease.
[11]
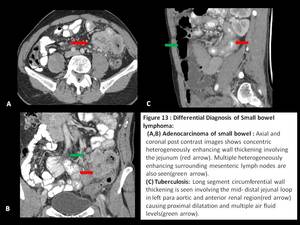
Fig. 13: Differential diagnosis of small bowel lymphoma
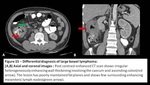
Large bowel:
Third most common site affected by GIT lymphoma with cecum and rectum being commonly affected sites.
Primary colonic lymphomas are extremely rare while secondary involvement following generalised disease process is commonly seen.[1,2,4]
Risk factors include immunosuppression (due to HIV infection or to immunosuppressive therapy after organ transplantation or another medical procedure) and inflammatory bowel disease.[4]
It can manifest as bulky polypoid growth,
infiltrative mass or aneurysmal dilatation.
As opposed to colorectal adenocarcinoma,
which is the main differential diagnosis,
lymphoma affects long segments and,
although it can narrow the lumen,
rarely can it cause obstruction.[2,3,4]
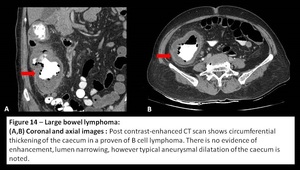
Fig. 14: Large bowel lymphoma
Differential diagnosis includes:
Adenocarcinoma of colon: Rectosigmoid junction is commonly affected part.
It appears as polypoidal or concentric soft tissue density mass that narrows the bowel lumen causes obstruction,
ulceration and fistula formation.
Varying degrees of local infiltration and surrounding lymphadenopathy may be seen.
[12]
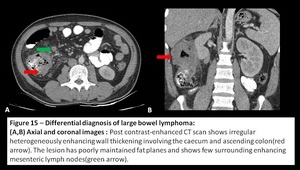
Fig. 15: Differential diagnosis of large bowel lymphoma
Peritoneal cavity:
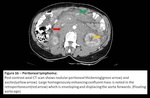
Peritoneal lymphomatosis is a rare clinical presentation that is often associated with high-grade primary gastrointestinal NHL and is radiologically indistinguishable from peritoneal carcinomatosis.[1,2] The patterns of involvement include discrete nodules,
a diffuse infiltrative mass,
and ascites.
Exudative ascites from peritoneal lymphomatosis shows high attenuation because of the increased proteinaceous content.[1,2]
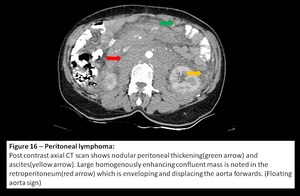
Fig. 16: Peritoneal lymphoma
Biliary Tract and Gall bladder:
Lymphomatous involvement of the biliary tree and gall bladder is rare.
Patterns of involvement of the gallbladder include an intraluminal polypoidal mass,
a large mass replacing the gallbladder,
and diffuse mural thickening.
The patterns of involvement of the bile ducts include a biliary stricture mimicking cholangiocarcinoma and a focal mass.
AIDS related lymphoma:
Lymphoma is the second most common neoplasm associated with AIDS.
AIDS-related lymphomas (ARL) have a number of highly unusual features when compared with non-ARL,
including involvement of the bone marrow and skin.[3]
In addition,
ARL have a striking predilection for extranodal areas of involvement,
and the GI tract is the most common extranodal site.
ARL may affect any abdominal organ,
most commonly LN,
the GI tract,
liver,
kidney,
adrenal gland,
omentum,
and abdominal wall.
Pattern of involvement in ARL is different from that of non ARL,
focal masses is the predominant type seen in solid and hollow organ ARL.[3]