Scrotal anatomy
The scrotal sac arises from the labioscrotal folds during embryonic development [1].
Around the 8th week of gestation,
medial to the inferior epigastric vessels a localized fold of peritoneum (the processus vaginalis) evaginates into the scrotal sac via the inguinal canal,
and with it carries all of the structures contained within the adult scrotal sac [1].
The testes descend caudally into the scrotum guided by the gubernaculum,
which extends from the inferior testicular pole to the labioscrotal fold,
under the influence of testosterone [2].
The processus vaginalis carries with it extensions of the abdominal fascia,
which give rise to the scrotal and spermatic cord layers eventually present from infancy [1,2].
By the end of the 1st year of life,
the scrotum will contain the testes intimately surrounded by the tunica albuginea,
a fine fibrous connective tissue layer that derives from mesenchymal cells [1].
Superficial to the tunica albuginea lies the tunica vaginalis,
which results from the closure of the processus vaginalis superiorly and contains a double layer of peritoneum lined by mesothelium [1].
The next outward layer is the internal spermatic fascia (derived from the transversalis fascia),
followed by the cremaster muscle and external spermatic fascia (derived from the internal oblique and external oblique muscles respectively) [1].
The scrotal skin and dartos muscle develop from the labioscrotal sac [1].
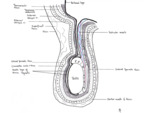
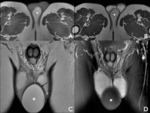
All these layers encase the spermatic cord (Fig.
1).
In practice,
these layers can be difficult to appreciate on imaging in the physiological state,
but pathology can help demonstrate some of these layers.
Adjacent to the testis lies the epididymis,
which contains between 15-20 efferent ductules connecting the testis to the vas deferens [1].
The epididymis is divided into three parts: the head (lying at the superior pole of the testis),
body and tail (at the lower pole of the testis) [1].
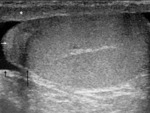
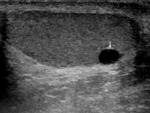
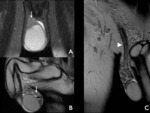
These structures can be easily identified on US and MRI (Fig.
2,
3,
4).
The vas deferens travels in the cephalad direction into the spermatic cord towards the seminal vesicles [1].
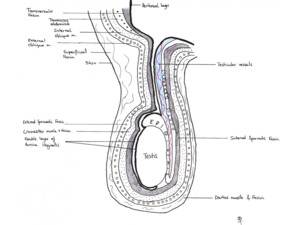
Fig. 1: Diagram of the adult scrotum demonstrating the layers of the spermatic cord, and the abdominal layers of which they are derived.
References: F. D. ALVES PEREIRA, Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016
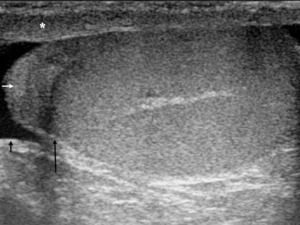
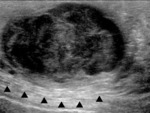
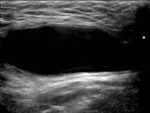
Fig. 2: Longitudinal section (LS) US image through the adult scrotum. The cremasteric muscle layer is demonstrated (*), with the linear hyperechoic external and internal spermatic fascia respectively immediately superficial and deep to it. The slightly loosened mixed echogenicity most superficial structure represents the skin. On the upper pole of the testis sits the epididymal head (short white arrow). The testis is enveloped by the two tunica vaginalis layers – parietal (short black arrow) and visceral (long black arrow).
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
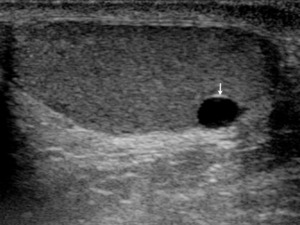
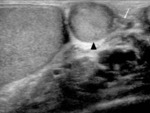
Fig. 3: LS US image through the left testis illustrating that certain scrotal structures may become more apparent in pathological cases. In this young adult man, the presence of a tunica albuginea cyst allows the tunica albuginea to be better appreciated (white arrow).
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
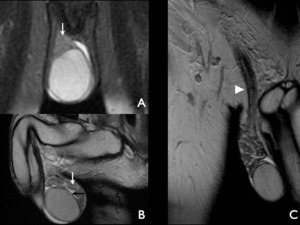
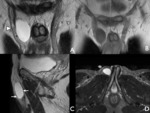
Fig. 4: Images A and B are T2-weighted, fluid sensitive, fat suppression sequences in the coronal and sagittal plane respectively. Image A demonstrated the typical relationship of the epididymal head and testis (white arrow). Image B shows the body and tail of the epididymis (white arrow). Image also illustrates how it is impossible to appreciate the tunica albuginea and vaginalis (visceral layer) as separate structures in the in the physiological state (black arrow). Image C is a T2-weighted coronal image through the anterior pelvis, which illustrates the course of the spermatic cord from the scrotum to the deep inguinal ring (white arrow head).
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
Spermatic cord leiomyosarcoma
Leiomyosarcomas are rare malignant neoplasms that develop from the spermatic cord [3].
Around one-tenth of all the sarcomas affecting the spermatic cord are leiomyosarcomas [3].
The most common solid tumours affecting the cord are lipomas; liposarcoma is the most common of the malignant neoplasms in the adult [3,4].
Epidemiology
It can affect individuals at any age,
but most men are over 40 years old [3].
Clinical features
Men typically present with a non-tender scrotal lump.
Imaging features
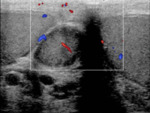
US - The US appearances are usually of a heterogeneous hypoechoic solid lesion emerging from the scrotal portion of the spermatic cord (Fig.
5).
These lesions are hypervascular and demonstrate colour flow (Fig.
6).
[3]
CT - This modality plays an important role in the staging of this neoplasm.
Although it provides limited detail about the lesion,
it may nonetheless demonstrate a soft tissue attenuation mass at the level of the scrotal portion of the spermatic cord (Fig.
7),
which enhances heterogeneously with contrast [3].
MRI - This imaging type is useful in assessing for local spread and in detecting local lymph nodes.
The typical appearances are of a soft tissue mass,
with T1 hypointense and T2 hyperintense signal [3].
Similarly to CT,
gadolinium administration results in heterogeneous enhancement.
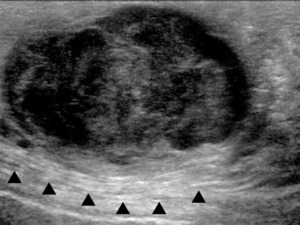
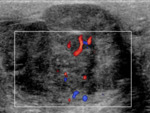
Fig. 5: LS US image through the right upper scrotum shows a lobulated disorganised mixed echogenicity lesion with close relationship to the echogenic spermatic cord. The borders are irregular and the internal structure poorly defined. This lesion was growing rapidly.
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
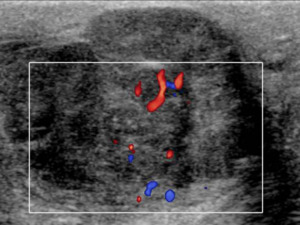
Fig. 6: TS US image through the right upper scrotum, once again shows the heterogeneous lesion, which demonstrates internal colour flow. This finding should always raise the suspicion of malignancy. The differential diagnosis for a lesion here would include a liposarcoma, leiomyosarcoma or angiosarcoma of the cord. This lesion was biopsied and shown to be a leiomyosarcoma.
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
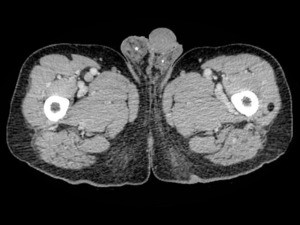
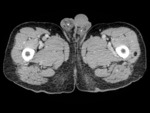
Fig. 7: Axial contrast-enhanced (portal venous phase) CT slice through the level of the proximal femur/ external genitalia of the same patient shown in Fig. 3. This shows the CT appearances of a proven case of leiomyosarcoma. It has a heterogeneous mixed attenuation appearance. Some of its elements show a similar attenuation to muscle (*), favouring the diagnosis of leiomyosarcoma, although a rhabdomyosarcoma is the differential diagnosis.
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
Scrotal lipoma
Lipomas are benign slow growing tumours which can occur anywhere in the body and are composed of adipose tissue.
It is the most common paratesticular neoplasm [1].
They can occur anywhere in the scrotum,
but most frequently arise from the spermatic cord [3].
Their size is highly variable.
Epidemiology
They can occur at any age and show no predilection for side.
Clinical features
Patients may present with a painless lump.
Clinically,
if large enough it may mimic polytestes.
Imaging features
US - In general,
lipomas of the spermatic cord are well defined,
classically hyperechoic and demonstrate no internal colour flow [5].
However,
these findings are neither sensitive nor specific and lipomas can show variable reflectivity [1],
may and can even be hypoechoic (Fig.
8).
For a definitive diagnosis,
many patients consequently undergo a MRI.
CT - CT is not usually the preferred method for evaluation of scrotal lipomas.
The typical characteristics of a lipoma on CT are that of a well-defined homogenous low attenuation mass,
with similar attenuation/ HU to that of subcutaneous fat [3].
MRI - Given that lipomas contain fat they are T1 and T2 hyperintense (Fig.
9) [1,3].
The requisite feature for confirmation of diagnosis on MRI is signal loss on frequency-selective fat saturated sequences (Fig.
9).
Another characteristic of lipomas on MRI is lack of enhancement following gadolinium administration.
[3]
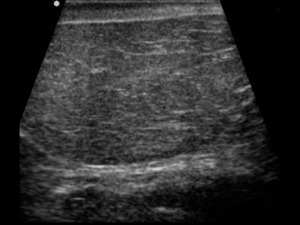
Fig. 8: TS US image through the midline of the scrotum demonstrates an encapsulated well defined lesion in a middle-aged man. The lesion was relatively hypoechoic and showed some internal septa. The diagnosis of a scrotal lipoma or possible liposarcoma was considered and the patient underwent a MRI for confirmation of the diagnosis.
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
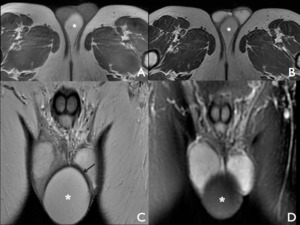
Fig. 9: Images A and B are T1 and T2 weighted axial slices through the upper femur/ external genitalia level, respectively. The lesion (*) is well defined and shows hyperintense T1 and T2 signal. mage C is a coronal slice of the anterior lower pelvis, which demonstrates the brim of T2 signal loss surrounding the lesion (arrow) consistent with the presence of a capsule, which can also be appreciated on image A. The lesion shows homogenous signal loss on the fat suppression, fluid sensitive coronal sequence (image D), confirming the diagnosis of a scrotal lipoma arising from the perineal fat, with no suspicious features.
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
Encysted hydrocele of the spermatic cord
Hydroceles of the spermatic cord are fluid collections that can occur along the spermatic cord [2,4].
They can be congenital or acquired.
The congenital presentation is due to failure of closure of the processus vaginalis; two types are recognized.
The encysted type,
where the fluid collection shows no communication with the tunica vaginalis inferiorly or the peritoneum superiorly [4].
In contrast,
the funicular type shows communication with the peritoneum via the internal inguinal ring [4].
Acquired encysted hydroceles may occur because of trauma,
infection or neoplasm [4].
Epidemiology
They are more common in children,
particularly the congenital type.
However,
acquired types may affect any age group [2].
Clinical features
Patients may present with a non-tender lump in the groin/ upper scrotum,
which on examination can be tense and may show fluctuation [2,6].
Imaging features
US - Encysted spermatic cord hydroceles are well defined generally completely anechoic structures (Fig.
10),
which tend to lie medially and anterior to the spermatic cord within the inguinal canal [4].
They are highly variable in size.
In the context of trauma,
the encysted collection may initially be heterogeneously hyperechoic,
and with time become hypoechoic [6].
CT - Encysted hydroceles of the spermatic cord may be diagnosed incidentally on CT.
This modality would again show a well-defined low (fluid) attenuation collection in the inguinal canal closely related to the spermatic cord [4].
MRI - This modality can be useful for further evaluation,
particularly if there is any concerning characteristics on US.
The encysted hydrocele demonstrates T1 and T2 hyperintense signal (Fig.
11),
with no enhancement following contrast administration with gadolinium [4].
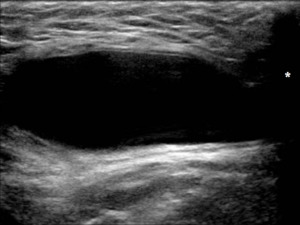
Fig. 10: LS US image through the right upper scrotum demonstrates a well-defined anechoic structure, which shows posterior acoustic enhancement and is adjacent to the right pubic tubercle superiorly (*). These appearances are typical of a cystic structure, in this case an encysted hydrocele of the spermatic cord. In a female patient this would be the typical location of a cyst of the canal of Nuck.
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
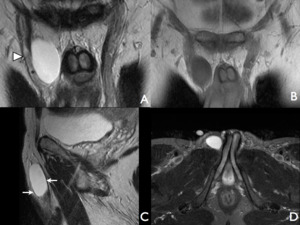
Fig. 11: Image A and B are coronal slices through the lower pelvis at the level of the external genitalia with T2 and T1 weighting respectively. The lesion lies medial and anterior to the spermatic cord (arrow head) and is T2 hyperintense and T1 hypointense, consistent with an encysted hydrocele of the spermatic cord. Image C demonstrates how the fluid collects between the anterior and posterior tunica vaginalis, showing no communication with peritoneum superiorly or inferiorly with the scrotal sac. The fluid sensitive sequence (D) confirms the presence of fluid collection along the spermatic cord in the axial plane.
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
Adenomatoid Tumour
Adenomatoid tumours are uncommon,
slow growing neoplasms,
which commonly affect the epididymis [1].
However,
they can occur in other areas such as the tunica albuginea and even the testis itself [1].
Their histological origin is controversial,
but the most recent studies suggest they have a mesothelial origin [7].
Epidemiology
They account for almost 33% of all the paratesticular neoplasms.
Although they may occur at any age,
they typically affect males in their twenties or above.
[7]
Clinical features
Patients may initially present with a scrotal solid lump and are usually asymptomatic,
although pain is reported in about one-third of patients.
In a minority of cases,
there may be a history of trauma and a small fraction also presents as epididymitis.
They are generally unilateral,
with a predilection for the left side.
[7]
Imaging features
US - There is a degree of variation in the sonographic appearances of these neoplasms and some characteristics may be non-specific.
The typical appearances may show an isoechoic lesion (Fig.
12) ranging from 0.5 to 5 cm in size at the head or tail of the epididymis (it occurs 4 times more commonly at the tail) [7].
Anechoic or hypoechoic cystic areas are not unusual.
Their location determines their morphology,
in the epididymis the lesion is commonly oval,
whereas in the testis it is often round [7].
Colour flow can be useful for further evaluation of these lesions,
which typically demonstrate flow (Fig.
13).
Another technique that may be useful when evaluating adenomatoid tumours is microbubble contrast enhanced ultrasound.
Administration of contrast results in tumour enhancement,
followed by early washout.
[8]
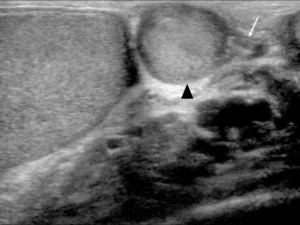
Fig. 12: TS US image through the lower right scrotum shows a round well defined lesion (arrow head) closely associated with the tail of the epididymis, which is relatively isoechoic to the epididymal tail (white arrow).
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.
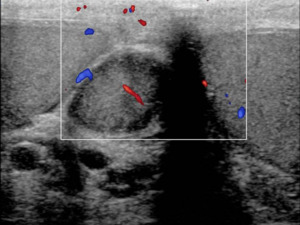
Fig. 13: TS US Doppler image at the same level as fig. 9a, demonstrating internal flow. These findings are typical for an adenomatoid tumour of the epididymis.
References: Department of Radiology, Norfolk and Norwich University Hospitals, Norwich, United Kingdom 2016.